 /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
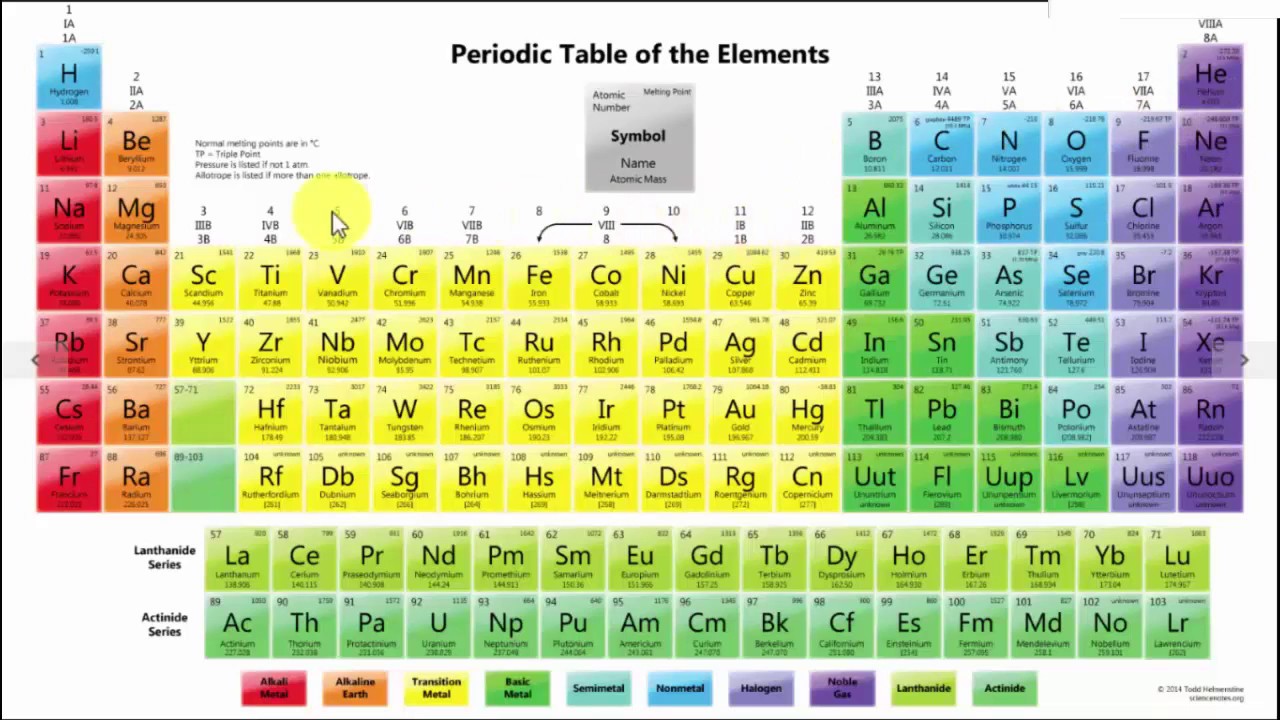
A visit to the Baku oilfields in 1863 began his long-term commitment to the emerging petrochemical industry, for example. In addition, he published an organic chemistry textbook and several articles for a technical encyclopaedia, as well as travelling widely in search of opportunities to apply scientific discoveries to Russia’s economic development. During the 1860s opinion shifted in his favour – fortunately for Mendeleev, as the regularities which pointed him towards the periodic table would have been less visible on the older system.Īfter returning to St Petersburg in 1861 Mendeleev resumed teaching at the university, while also lecturing at the city’s Technological Institute. But at Karlsruhe Cannizzarro revived the ideas of his fellow-countryman Amadeo Avogadro to support the H 2O water formula, and an atomic weight of 16 for oxygen. Others preferred a system based on an atomic weight of eight for oxygen, assuming that water’s formula was HO, rather than H 2O. Some chemists claimed these weights were irrelevant, or denied the physical existence of atoms altogether. 
This was a crucial step towards the periodic system, as previously there had been considerable dispute over the assigning of atomic weights to the elements. In 1860 he attended the Karlsruhe conference, where the Italian chemist Stanislau Cannizzaro delivered a ground-breaking paper on atomic weights (now called relative atomic masses). Establishing a careerĪt Heidelberg University in Gemany, Mendeleev did research on several topics, including surface tension, capillarity and evaporation, and he retained an interest in intermolecular forces throughout his career. In 1859 he received state funding for two years of advanced study abroad. Shortly afterwards the University of St Petersburg licensed him as a chemistry tutor, allowing him access to its laboratory. In the autumn of 1856 Mendeleev successfully defended a master’s thesis on relationships between the specific volumes of substances and their crystallographic and chemical properties. However, a year teaching in the more benign climate of the Crimea improved his health significantly, and a new doctor confidently dismissed the previous diagnosis. By then both his mother and sister had died, and he himself was suffering from what appeared to be tuberculosis. 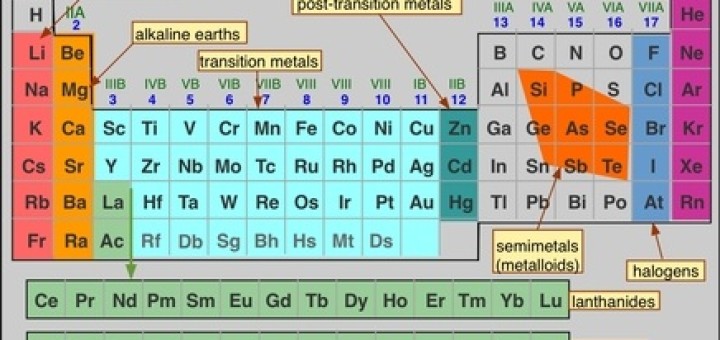
Further articles for scientific and technical periodicals followed, but he lacked a secure income. He graduated in 1855, and his dissertation – on isomorphism and other relationships between physical form and chemical composition – was published in a mining journal. A lecturer there – Alexander Voskresensky, who had studied in Germany under Justus Liebig – encouraged Dmitri’s interest in chemistry. When this plan failed, they moved to St Petersburg and in 1850 Dmitri was accepted (somewhat reluctantly) by the college where his father had trained as a teacher. Then Ivan died, and in 1849 Maria took her two youngest children to Moscow, hoping that her brother would help Dmitri enter university there. This enterprise supported the family until 1848, when it burnt down. The inadequacy of Ivan’s pension drove his wife Maria to take on the management of a semi-derelict glass-works, formerly run by her brother. He was the youngest of more than a dozen Mendeleev siblings, and soon after his birth in 1834 ill-health forced his father Ivan, a high-school teacher, into retirement. His home, Tobolsk, is 1000km nearer to Beijing than to Paris, and his pathway from there to scientific eminence was difficult. Dmitri Mendeleev taught in St Petersburg and wrote his own textbooks – it was this process that lead to his periodic discoveryĭmitri grew up in Siberia, on the outer edge of western civilisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
